Clinical Decision Support (CDS) systems have been in hospitals for decades. The first generation was simple: if the doctor orders Drug A and the patient is on Drug B, show a pop-up. The second generation added risk scores — logistic regression models predicting sepsis, readmission, or acute kidney injury.
Now we are in the third generation: LLM-powered agents that can reason over unstructured clinical notes, synthesize medical literature, and generate context-aware recommendations.
They layered on top. The best CDS systems in 2026 combine all three: deterministic rules for drug interactions and dosage checks (100% precision, zero tolerance for error), ML models for risk prediction (probabilistic but data-driven), and LLM agents for complex reasoning tasks (flexible but requiring safety guardrails). Understanding when to use which — and how to integrate them — is the engineering challenge this guide addresses.
According to a 2023 Health Affairs study, hospitals with mature CDS implementations see 15-20% reductions in adverse drug events and 8-12% reductions in unnecessary diagnostic testing. But 60% of CDS alerts are overridden by clinicians. The technology works — the implementation usually does not. This guide covers how to get both right.
Three Types of CDS: Rules, Models, and Agents
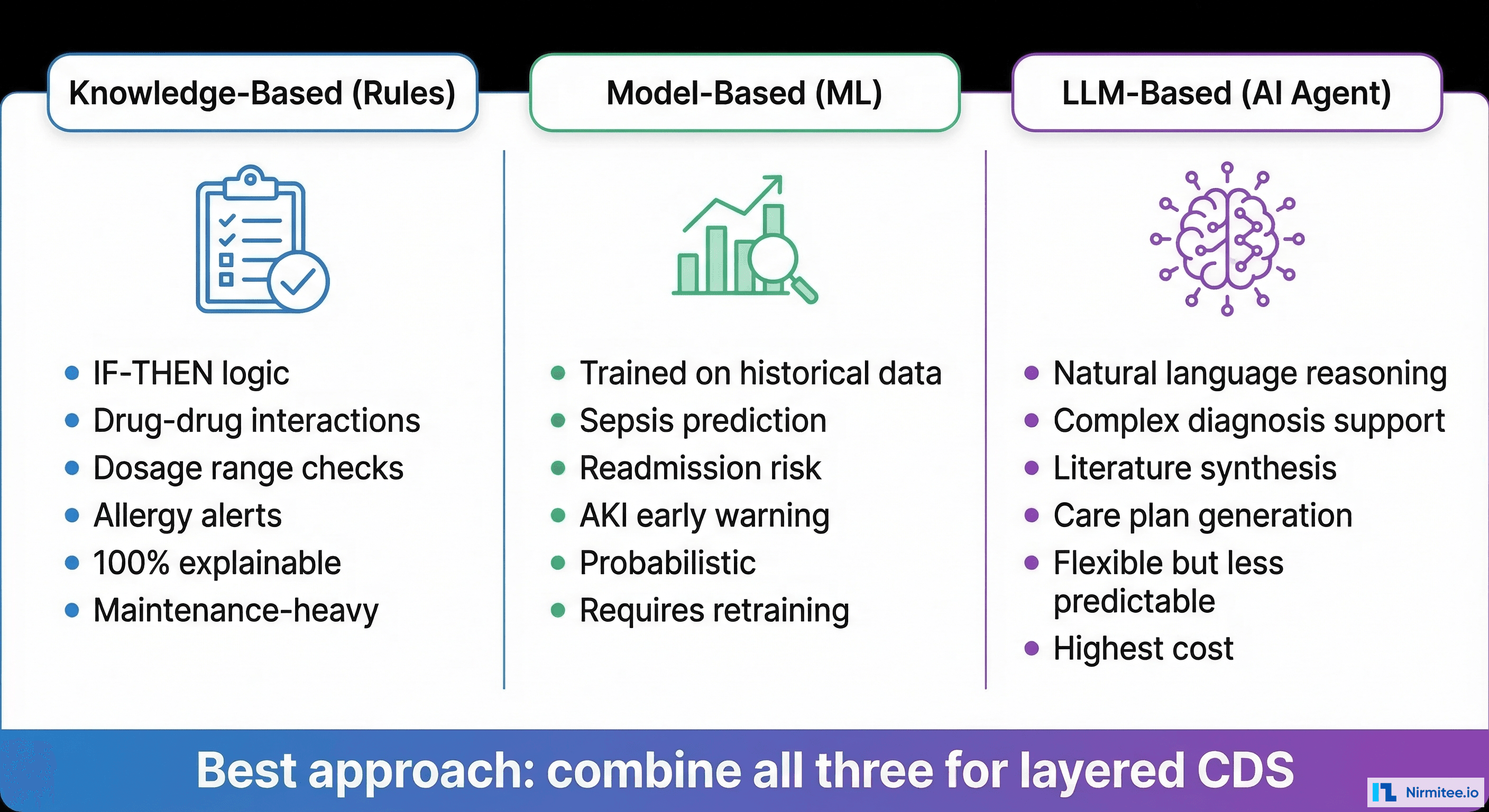
Type 1: Knowledge-Based CDS (Rules Engines)
IF-THEN logic operating on structured clinical data. Drug-drug interaction checking, allergy alerting, dosage range validation, and duplicate order detection. These are the workhorses of clinical decision support — unglamorous but responsible for preventing the majority of medication errors in US hospitals.
Implementation: Most EHR platforms (Epic, Cerner/Oracle Health, MEDITECH) include built-in rules engines. External CDS knowledge bases like First Databank (FDB) and Medi-Span provide drug interaction databases that power these rules. You do not build drug interaction rules from scratch — you license a clinical knowledge base and integrate it.
Strengths: 100% explainable. Deterministic — same input always produces the same output. Zero latency (millisecond response times). No training data required. The regulatory pathway is clear (rules are reviewable by clinical committees).
Weaknesses: Cannot handle nuance or context. A rules engine cannot distinguish between a 25-year-old athlete with a heart rate of 50 bpm (normal) and a 75-year-old sedentary patient with a heart rate of 50 bpm (potentially bradycardic). It fires the same alert for both because it only evaluates discrete thresholds.
Type 2: Model-Based CDS (Machine Learning)
Trained on historical patient data to predict clinical outcomes. Sepsis prediction (using vital signs, lab trends, and nursing assessments), AKI early warning (creatinine trajectories and fluid balance), readmission risk (discharge diagnoses, social determinants, prior utilization), and falls risk (mobility scores, medication profiles, age).
Implementation: Models are typically trained offline on de-identified historical data, validated by clinical teams, and deployed as microservices that the EHR calls via CDS Hooks or internal APIs. The data de-identification pipeline and FHIR-based data extraction are prerequisites that most teams underestimate.
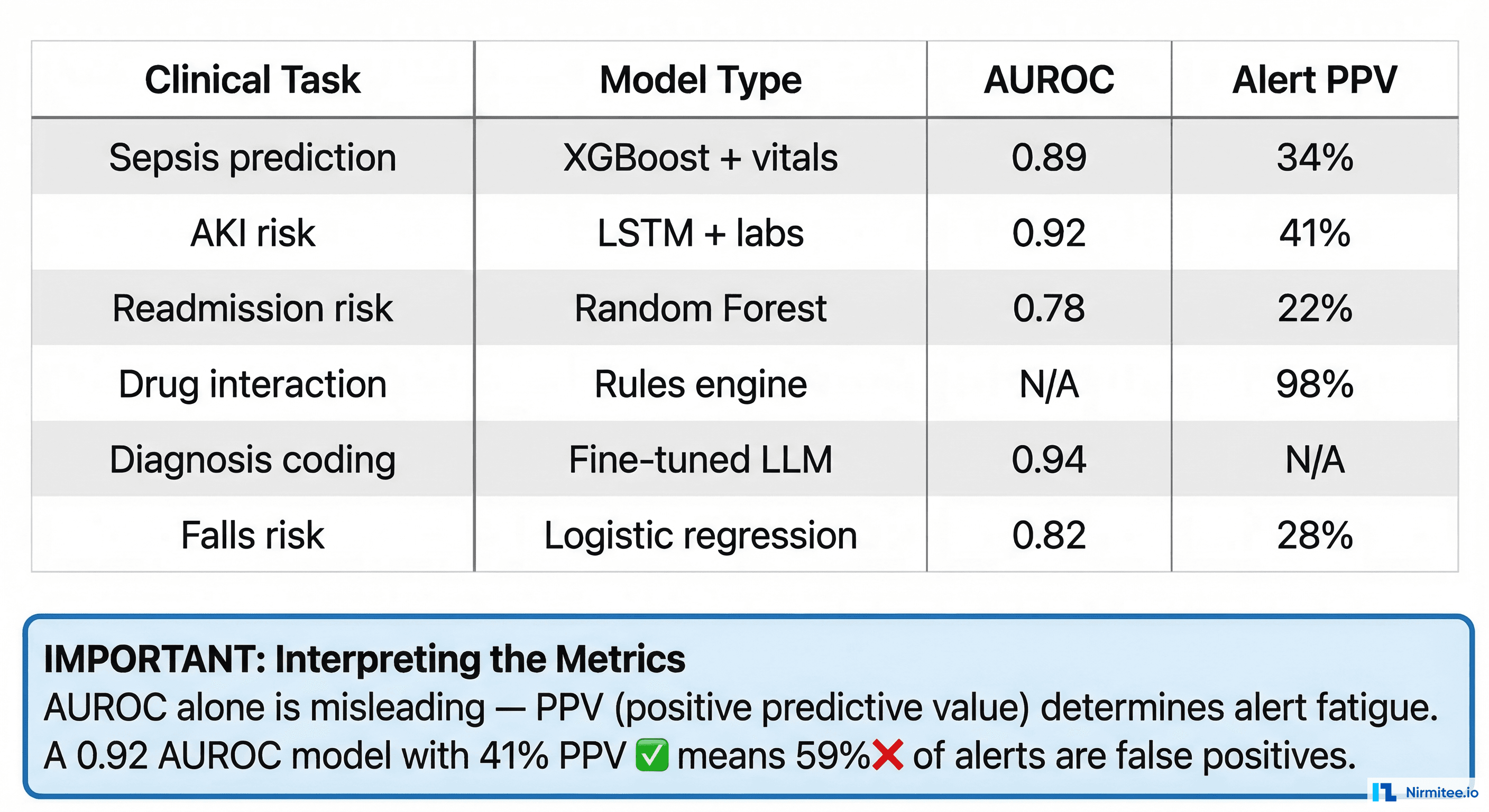
Strengths: Captures patterns invisible to rules engines. Can integrate dozens of variables simultaneously. Improves with more data. Clinical evidence that ML-based sepsis prediction reduces mortality by 18-25% when implemented correctly (Adams et al., Nature Medicine 2022).
Weaknesses: Probabilistic — false positives cause alert fatigue. Requires ongoing retraining as clinical practice changes (model drift). Less explainable than rules (though SHAP/LIME help). The FDA regulatory pathway is more complex for higher-risk applications.
Type 3: LLM-Based CDS (AI Agents)
Large language models that reason over unstructured clinical text, synthesize medical literature, and generate contextual recommendations. Use cases: differential diagnosis support (given symptoms and test results, suggest possible diagnoses with reasoning), care plan generation (given a complex patient, draft a multi-disciplinary care plan), clinical documentation assistance (extract structured data from free-text notes), and EHR-integrated decision support via CDS Hooks.
Strengths: Handles unstructured data that rules and ML models cannot process. Generates human-readable explanations. Can synthesize information from multiple sources (patient record + clinical guidelines + medical literature). Adapts to novel situations without retraining.
Weaknesses: Hallucination risk in clinical contexts is a patient safety concern. Cost per interaction is 50-100x higher than rules or ML models. Latency (2-10 seconds) may be unacceptable for real-time alerting. Safety guardrails are essential and add engineering complexity.
The Five Pillars of Effective CDS

The ONC's CDS Five Rights framework defines the standard for effective clinical decision support. Every CDS implementation should be evaluated against these criteria:
- Right Information: The recommendation is clinically accurate, evidence-based, and relevant to the patient's specific context. A drug interaction alert for a medication the patient discontinued 6 months ago is incorrect.
- Right Person: The alert reaches the clinician who can act on it. A pharmacist-level drug interaction alert shown to a scheduling clerk is the wrong person. Role-based alert routing is essential.
- Right Channel: The recommendation is delivered through the appropriate mechanism — a passive info button vs. an interruptive alert vs. an order set suggestion. Not everything needs a pop-up.
- Right Time: The alert fires when the clinician can act on it. A sepsis alert that fires 4 hours after the patient has already deteriorated is the wrong time. Real-time data pipelines (FHIR Subscriptions) are often required.
- Right Format: The recommendation is presented in a way that supports rapid decision-making. A 500-word paragraph explaining a drug interaction is the wrong format. A structured card with severity, affected drugs, recommended action, and evidence link is the right format.
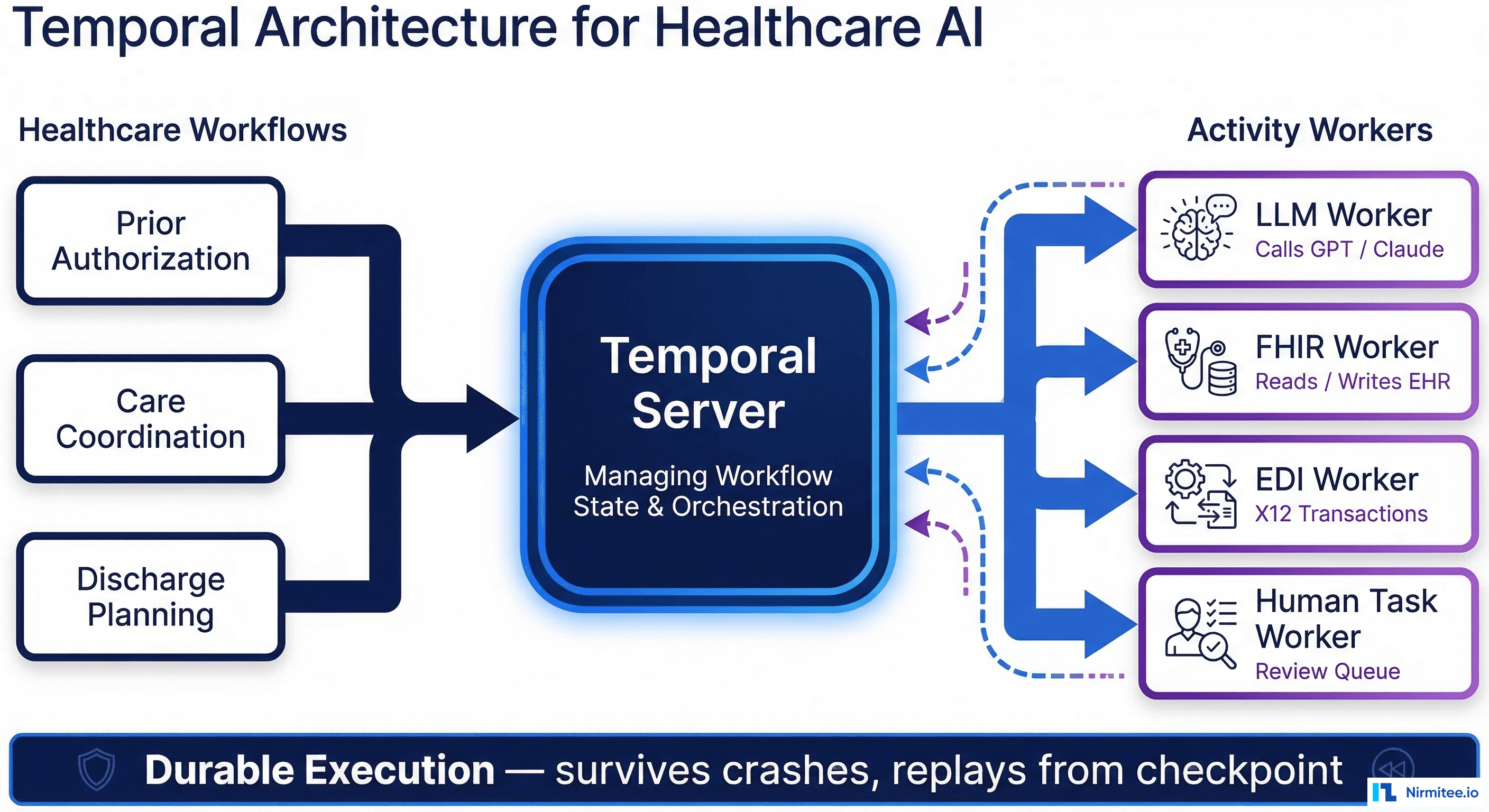
CDS Hooks: The Integration Standard
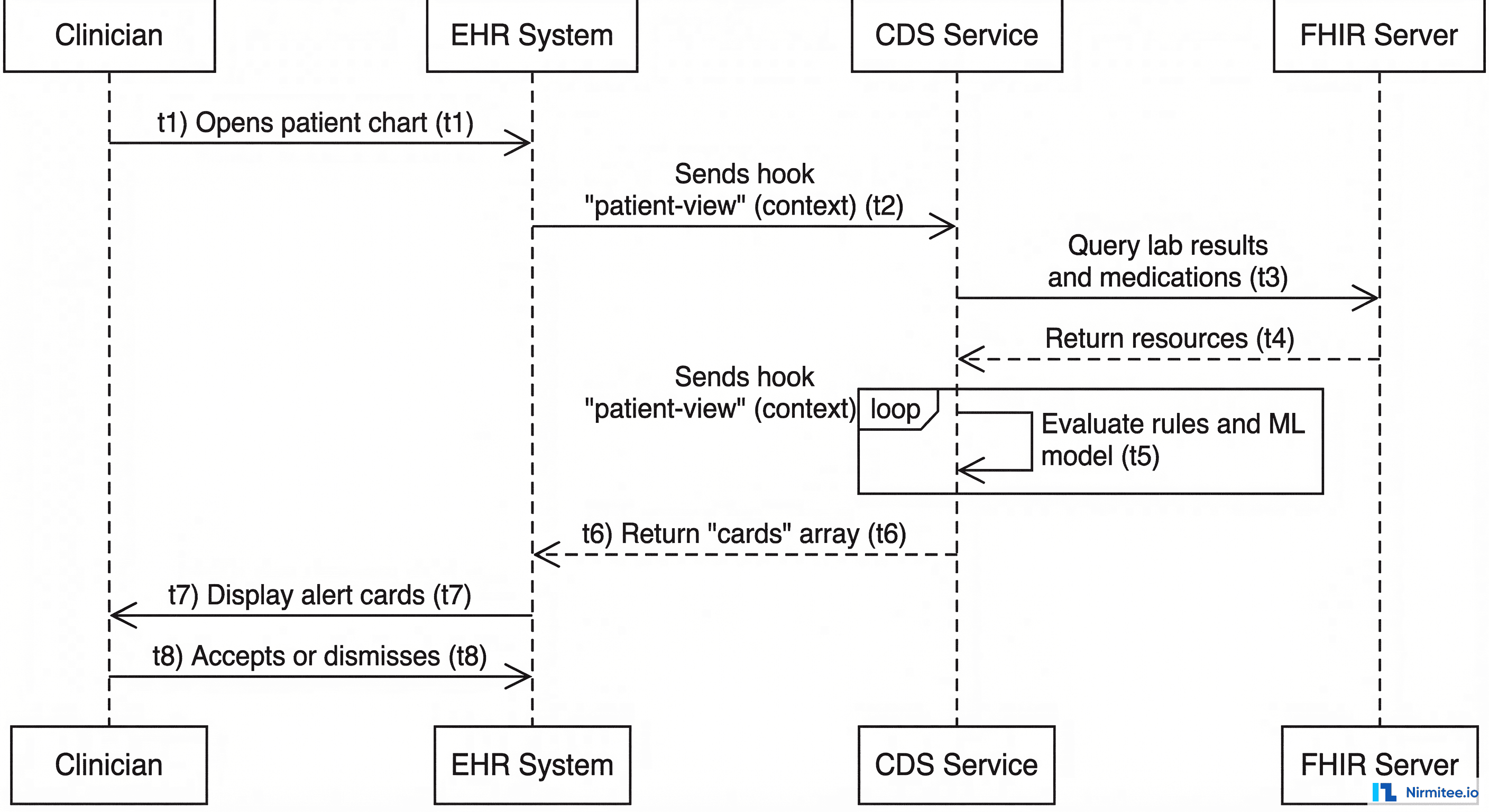
CDS Hooks is the HL7-published standard for integrating CDS services with EHR systems. It defines a REST-based API where the EHR sends clinical context to external CDS services at defined trigger points (hooks), and the CDS service returns structured recommendation cards.
How It Works
When a clinician performs a specific action in the EHR (opening a patient chart, signing an order, prescribing a medication), the EHR fires a hook to registered CDS services:
POST https://cds.example.org/cds-services/sepsis-risk
Content-Type: application/json
{
"hookInstance": "d1577c69-dfbe-44ad-bd63-3c5194a67584",
"hook": "patient-view",
"fhirServer": "https://ehr.example.org/fhir",
"fhirAuthorization": {
"access_token": "eyJhbG...",
"token_type": "Bearer",
"scope": "patient/*.read"
},
"context": {
"userId": "Practitioner/dr-smith-4821",
"patientId": "Patient/p-28491",
"encounterId": "Encounter/enc-77832"
}
}The CDS service queries the FHIR server for relevant patient data (vitals, labs, medications), runs its decision logic (rules, ML model, or LLM), and returns recommendation cards:
{
"cards": [
{
"uuid": "card-sepsis-risk-001",
"summary": "Elevated sepsis risk: qSOFA score 2/3",
"detail": "Patient has 2 of 3 qSOFA criteria: respiratory rate 24 (>=22), systolic BP 95 (<100). Consider blood cultures and lactate level.",
"indicator": "warning",
"source": {
"label": "Sepsis Early Warning System",
"url": "https://cds.example.org/evidence/sepsis-qsofa"
},
"suggestions": [
{
"label": "Order blood cultures and serum lactate",
"actions": [
{
"type": "create",
"description": "Blood culture order",
"resource": {
"resourceType": "ServiceRequest",
"status": "draft",
"intent": "proposal",
"code": {
"coding": [{
"system": "http://loinc.org",
"code": "600-7",
"display": "Blood culture"
}]
}
}
}
]
}
]
}
]
}The card includes a pre-built FHIR ServiceRequest that the clinician can accept with one click — the order is created directly in the EHR. This "suggestion with action" pattern dramatically increases CDS adoption because the clinician's effort to follow the recommendation is near zero.
Supported Hooks in Major EHRs
| Hook | Trigger | Epic | Oracle Health | MEDITECH |
|---|---|---|---|---|
| patient-view | Clinician opens patient chart | Yes | Yes | Partial |
| order-select | Clinician selects an order | Yes | Yes | No |
| order-sign | Clinician signs/submits orders | Yes | Yes | Partial |
| medication-prescribe | Clinician prescribes medication | Yes | Limited | No |
| encounter-discharge | Patient discharged | Yes | No | No |
Epic has the most comprehensive CDS Hooks support. Oracle Health (formerly Cerner) supports the core hooks but has gaps in medication-prescribing workflows. MEDITECH support is limited and often requires Mirth Connect as a middleware layer to bridge the gap.
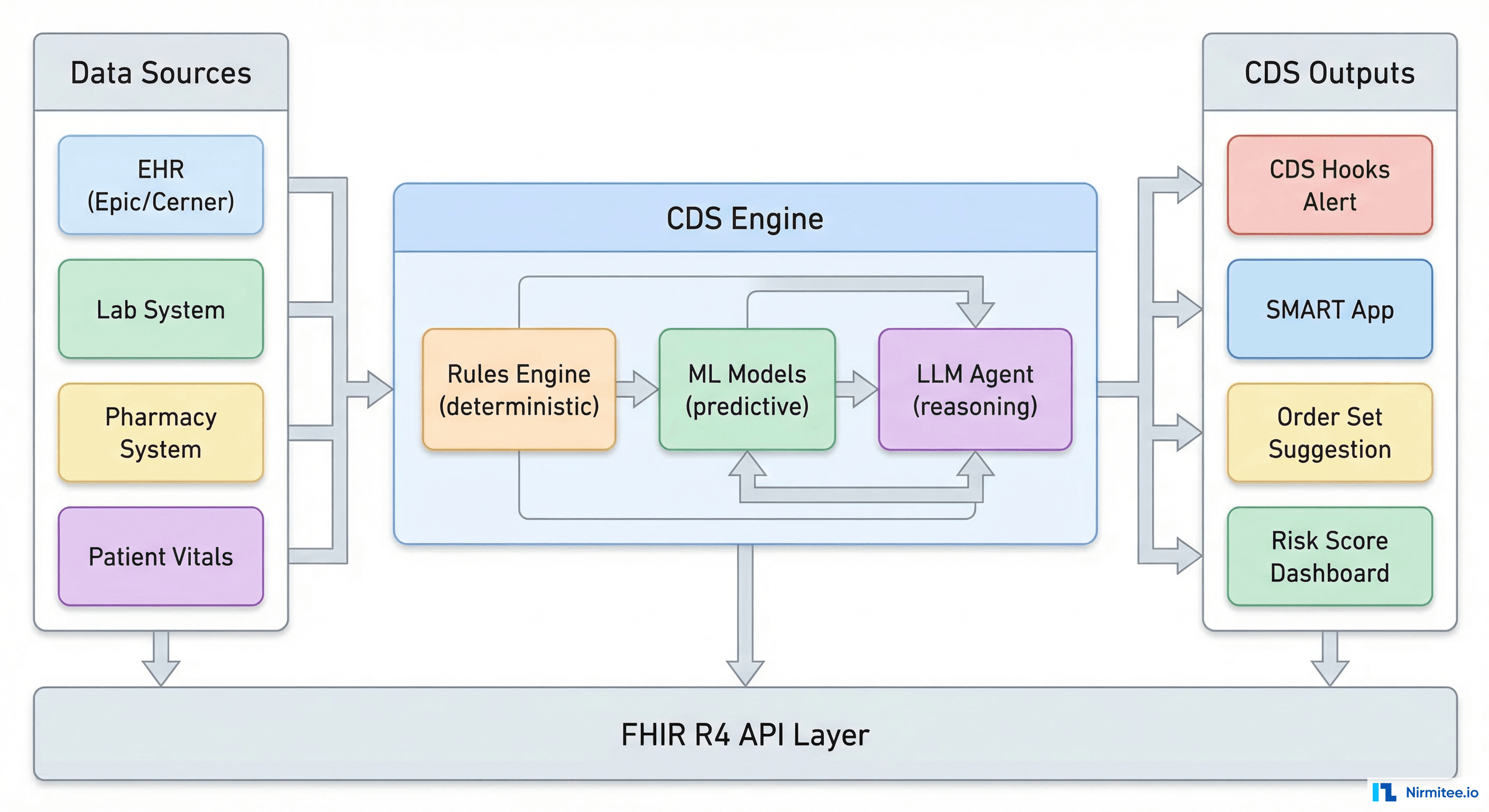
Architecture: Designing a Production CDS System
A production CDS system is not a single model behind an API. It is a layered architecture that combines multiple decision types, manages clinical knowledge, handles alert routing, and provides the audit and explainability infrastructure that compliance teams require.
Data Layer
Your CDS system needs real-time access to patient data. This means either direct FHIR API queries to the EHR (adds latency per CDS request) or a synchronized clinical data store that receives real-time updates via HL7v2 ADT/ORU messages or FHIR Subscriptions. Most production systems use the synchronized store approach — querying the live EHR for every CDS request adds 200-500ms of latency that makes real-time alerting impractical.
Decision Layer
Route each clinical question to the appropriate decision type:
- Drug interactions, allergies, dosage checks: Rules engine. Zero tolerance for error — deterministic logic with licensed knowledge bases.
- Sepsis risk, AKI prediction, readmission probability: ML models. Deploy as microservices, each with its own FHIR data extraction, feature engineering, and prediction endpoint.
- Differential diagnosis, care plan synthesis, complex medication decisions: LLM agent with safety guardrails. Always with human-in-the-loop review for clinical actions.
Alert Management Layer
This is where most CDS implementations fail. Without alert management, you get alert fatigue — and alert fatigue kills CDS adoption.
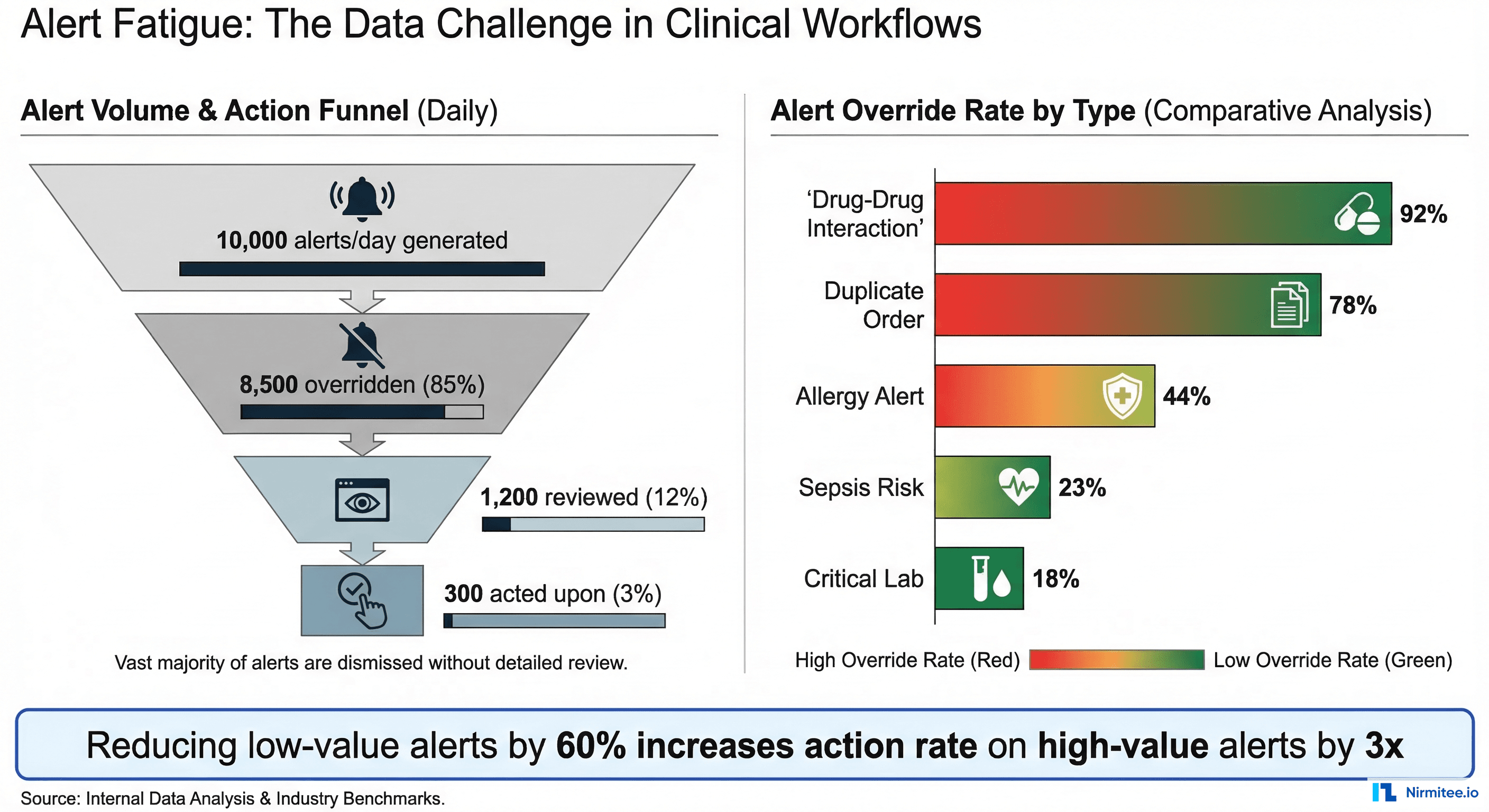
The data is stark: a 2019 study in JAMIA found that clinicians override 49-96% of drug-drug interaction alerts depending on severity category. A 2024 AMIA analysis of 12 health systems found that reducing low-value alerts by 60% increased clinician action rates on remaining alerts from 8% to 24% — a 3x improvement.
Alert management strategies that work:
- Severity tiering: Only interruptive alerts (pop-ups that block workflow) for high-severity, high-confidence findings. Everything else goes to passive displays (info panels, dashboard flags).
- Context suppression: If the patient has been on Drug A and Drug B together for 6+ months with no adverse events, suppress the interaction alert on subsequent encounters. The clinician and patient have already accepted this risk.
- Role-based routing: Pharmacist-level alerts go to pharmacists. Nursing assessments go to nurses. Diagnostic suggestions go to physicians. Stop routing everything to everyone.
- Frequency capping: The same alert for the same patient should not fire more than once per encounter unless clinical data has changed.
Regulatory Considerations: When CDS Becomes a Medical Device
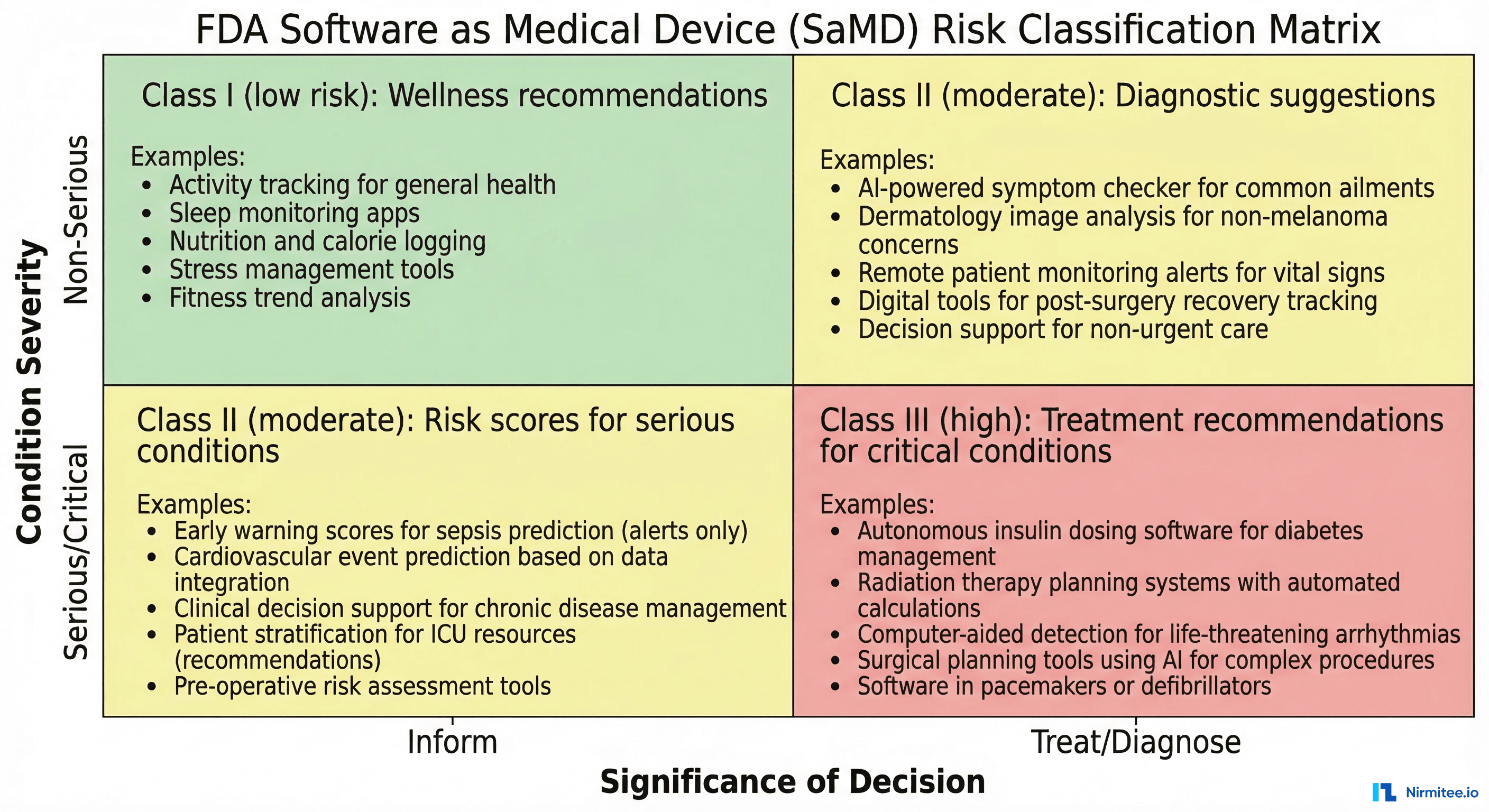
Not all CDS requires FDA clearance — but the line is moving. The FDA's framework for AI/ML-based Software as a Medical Device (SaMD) classifies CDS based on two dimensions: the significance of the information provided and the severity of the healthcare situation.
CDS That Is Exempt from FDA Oversight (Section 3060 of 21st Century Cures Act)
- Displays or summarizes patient data without recommendations
- Provides clinical knowledge (reference information, guidelines) not specific to an individual patient
- Supports administrative or operational decisions (not clinical)
- The clinician independently reviews the basis for the recommendation (the system shows its reasoning, not just the conclusion)
CDS That Requires FDA Clearance
- Acquires, processes, or analyzes a medical image (radiology AI)
- Provides a specific diagnosis or treatment recommendation without showing reasoning
- Is intended to replace clinical judgment rather than support it
- Processes data from in vitro diagnostic devices
The critical distinction for LLM-based CDS: if your system shows the evidence and reasoning alongside the recommendation (allowing the clinician to independently evaluate it), it likely qualifies for the Cures Act exemption. If it presents a recommendation as a black-box conclusion ("prescribe Drug X"), it is likely a medical device requiring FDA clearance.
Building Your First CDS Service: A Practical Checklist
- Start with rules, not ML. Implement drug interaction checking and allergy alerting first. These are well-defined, clinically validated, and deliver immediate value with zero model risk.
- Pick one ML use case. Sepsis prediction is the most common starting point — well-studied, high clinical impact, and validated models exist. Do not attempt 5 ML models simultaneously.
- Implement CDS Hooks. This is your integration pathway. Build one CDS Hooks service for the
patient-viewhook, deploy it, and validate the end-to-end flow before adding complexity. - Measure alert acceptance rate from day one. If clinicians override more than 70% of your alerts in the first month, you have an alert quality problem, not an adoption problem. Fix the alerts.
- Build the audit trail. Every alert generated, every clinician action (accepted, overridden, modified), and every data input that triggered the alert. This is your HIPAA compliance evidence and your CDS quality improvement dataset.
- Add LLM-based CDS last. Only after rules and ML models are running reliably. LLM agents add value for complex reasoning tasks, but they require the most engineering investment in safety guardrails, testing infrastructure, and monitoring.
AI in healthcare demands both technical depth and domain expertise. See how our Healthcare AI Solutions team can help you ship responsibly. We also offer specialized Healthcare Software Product Development services. Talk to our team to get started.
Frequently Asked Questions
What is the difference between CDS and CDSS?
CDS (Clinical Decision Support) is the general category. CDSS (Clinical Decision Support System) refers to a specific implemented system. In practice, the terms are used interchangeably. The HL7 standard uses "CDS" — we recommend following that convention.
Do I need FDA clearance for my CDS system?
It depends on what your system does and how it presents information. CDS that displays patient data, provides reference information, or shows reasoning alongside recommendations is generally exempt under the 21st Century Cures Act. CDS that provides autonomous diagnostic or treatment recommendations without showing reasoning likely requires FDA 510(k) or De Novo clearance. Consult regulatory counsel for your specific use case.
How do I reduce alert fatigue in my CDS system?
Three strategies with the most evidence: (1) suppress alerts for combinations the patient has tolerated for 6+ months, (2) tier alerts by severity and only make high-severity alerts interruptive, (3) route alerts to the appropriate role (pharmacist alerts to pharmacists, not all clinicians). Most organizations can reduce alert volume by 40-60% without losing any clinically significant alerts.
Which EHR has the best CDS Hooks support?
Epic, followed by Oracle Health. Epic supports patient-view, order-select, order-sign, medication-prescribe, and encounter-discharge hooks. Oracle Health supports the core hooks but has gaps in medication workflows. MEDITECH has limited native support and typically requires middleware like Mirth Connect to enable CDS Hooks integration.
What does a CDS implementation cost?
Rules-based CDS (drug interactions, allergies): $50K-150K, including knowledge base licensing and EHR configuration. ML-based CDS (single prediction model): $150K-400K including model development, validation, and deployment infrastructure. LLM-based CDS (agent with guardrails): $300K-800K including safety engineering, compliance review, and ongoing monitoring. These ranges assume an existing FHIR infrastructure — if you need to build the data layer first, add 30-50%.
Designing effective CDS is equal parts clinical knowledge, software engineering, and human factors research. At Nirmitee, we build healthcare AI systems where the decision support, EHR integration, and safety infrastructure work together from day one. If you are planning a CDS implementation — whether rules-based, ML-driven, or LLM-powered — talk to our team. We will help you avoid the alert fatigue trap and build CDS that clinicians actually use.