A year ago, every healthtech conference had the same prediction: AI would transform clinical workflows by the end of 2025. We wrote our original analysis of key trends to watch. Now, with a full year of data, we can measure what actually happened.
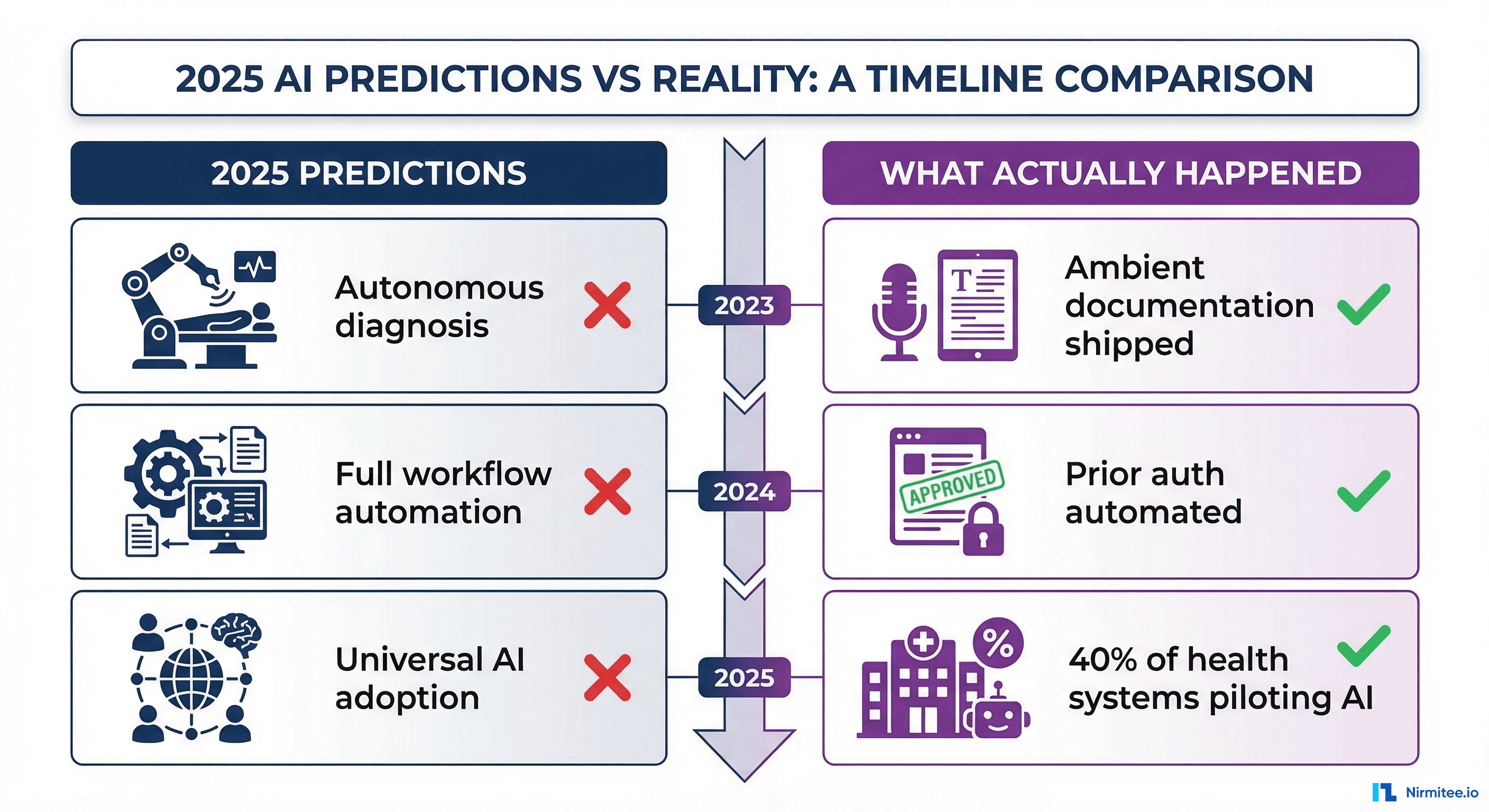
Predictions That Landed
| Prediction | What Happened | Evidence |
|---|---|---|
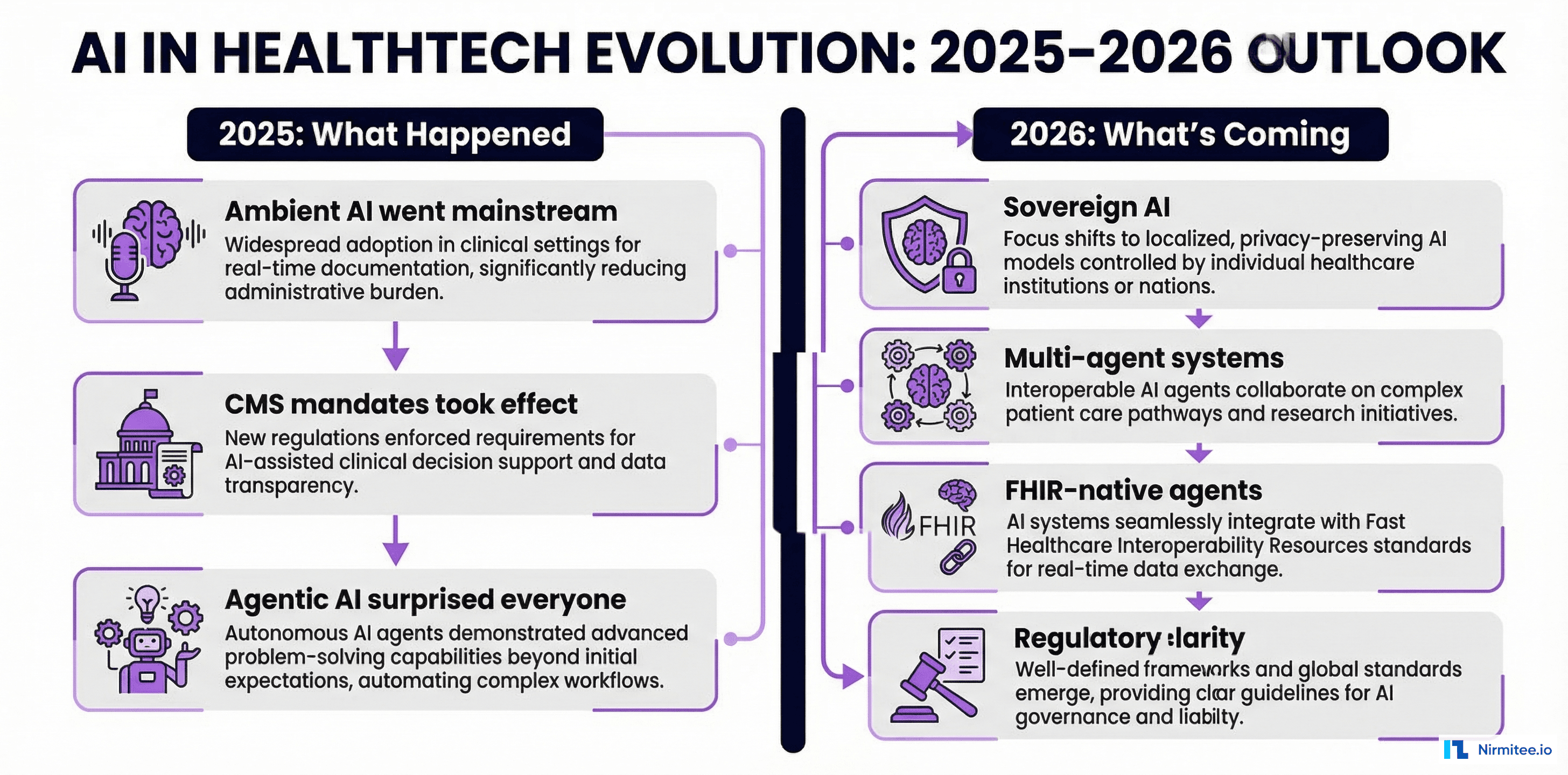
| Ambient documentation goes mainstream | Shipped by DAX Copilot, Abridge, Suki at scale | 70% documentation time reduction reported across 200+ health systems |
| Prior authorization gets automated | Cohere Health processes 20M+ decisions annually | 70% auto-approval rate, sub-30-second processing |
| CDS Hooks adoption accelerates | Epic and Oracle Health expanded CDS Hooks support | HL7 CDS Hooks STU 2.0 published with broader hook types |
| FHIR R4 becomes the baseline | ONC USCDI v3 mandated FHIR R4 for certified health IT | 95%+ of certified EHRs now support FHIR R4 APIs |
Predictions That Missed
| Prediction | What Actually Happened | Why |
|---|---|---|
| Autonomous clinical decision-making | AI remains advisory, not autonomous | FDA and clinical risk tolerance prevent autonomous decisions. CDS suggestions require clinician confirmation. |
| Universal AI adoption across health systems | 40% of health systems have AI pilots, 15% in production | Integration complexity and change management slow adoption. Most systems are still building FHIR infrastructure. |
| AI replacing coding and billing staff | AI augmenting coders, not replacing them | Revenue cycle AI improves accuracy but requires human oversight for complex cases and appeals. |
The Five Trends Defining Healthcare AI in 2026

Trend 1: Sovereign Healthcare AI
National governments are asserting control over how healthcare AI is developed, trained, and deployed within their borders. This is not theoretical policy — it is shipping legislation with enforcement mechanisms.
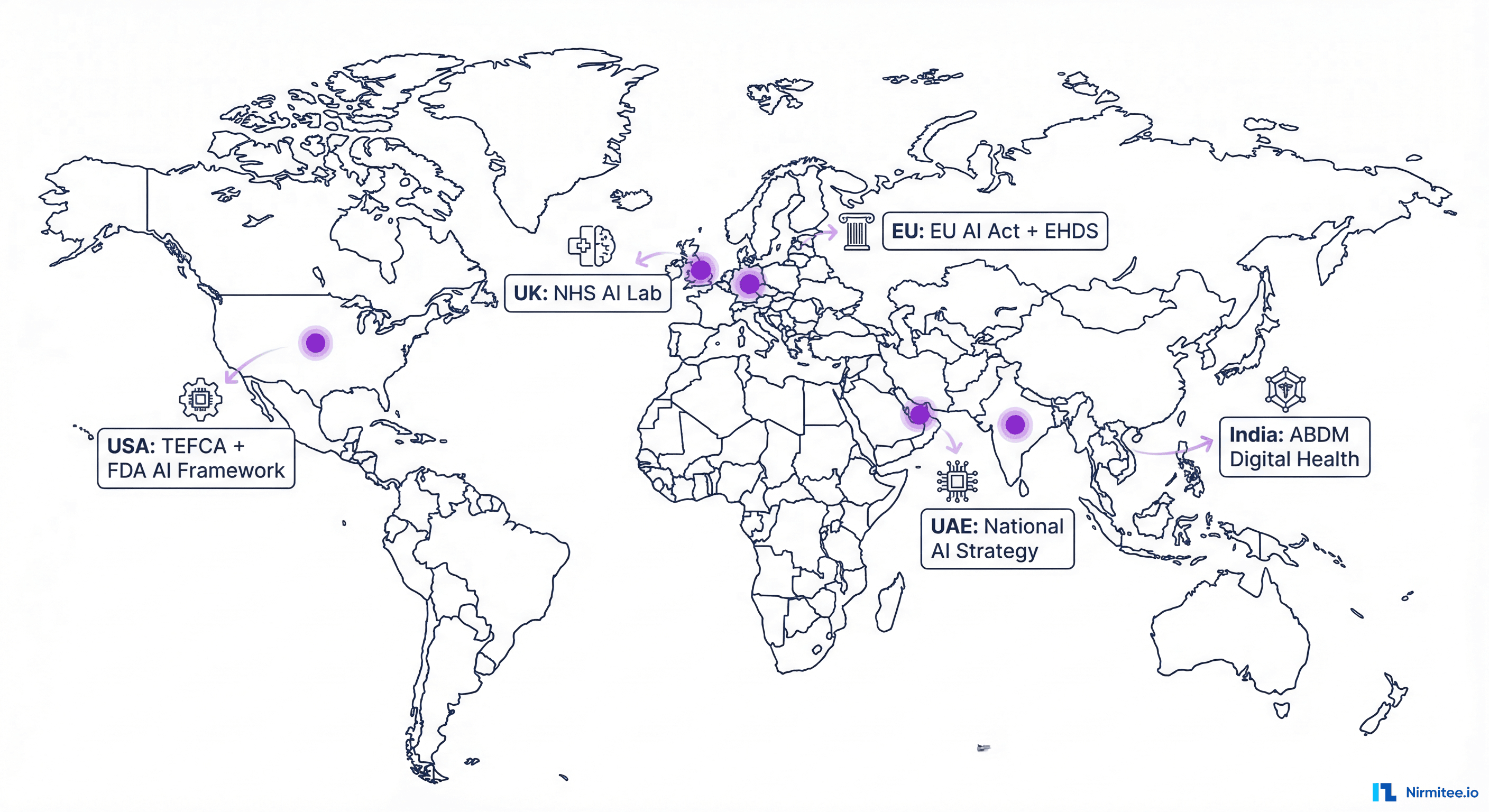
- European Union — The EU AI Act classifies healthcare AI systems as "high-risk," requiring conformity assessments, ongoing monitoring, and transparency documentation before deployment. The European Health Data Space (EHDS) creates a framework for secondary use of health data for AI training with strict governance.
- United States — The ONC's HTI-2 final rule, expected in 2026, includes requirements for AI transparency in certified health IT. The FDA is developing a regulatory framework for continuously learning AI/ML devices through the Predetermined Change Control Plan.
- India — The ABDM (Ayushman Bharat Digital Mission) is building the national health data infrastructure that AI systems will consume. The DPDP Act (Digital Personal Data Protection) establishes consent requirements for health data use in AI training.
- United Kingdom — NHS AI Lab is funding real-world deployments and building a national AI evaluation framework. The UK's regulatory sandbox approach moves faster than the EU's prescriptive framework.
- Gulf States — UAE's national AI strategy targets healthcare as a priority vertical, with government-funded AI research centers and fast-track regulatory pathways.
For healthtech companies, the implication is clear: you cannot build one global healthcare AI product. Each market requires compliance with local data residency, training data governance, and regulatory approval frameworks.
Trend 2: Multi-Agent Clinical Systems
Single-purpose AI tools are giving way to multi-agent systems where specialized AI agents collaborate on complex clinical workflows. Instead of one monolithic AI, you have an orchestrated team of agents, each expert in a specific domain.
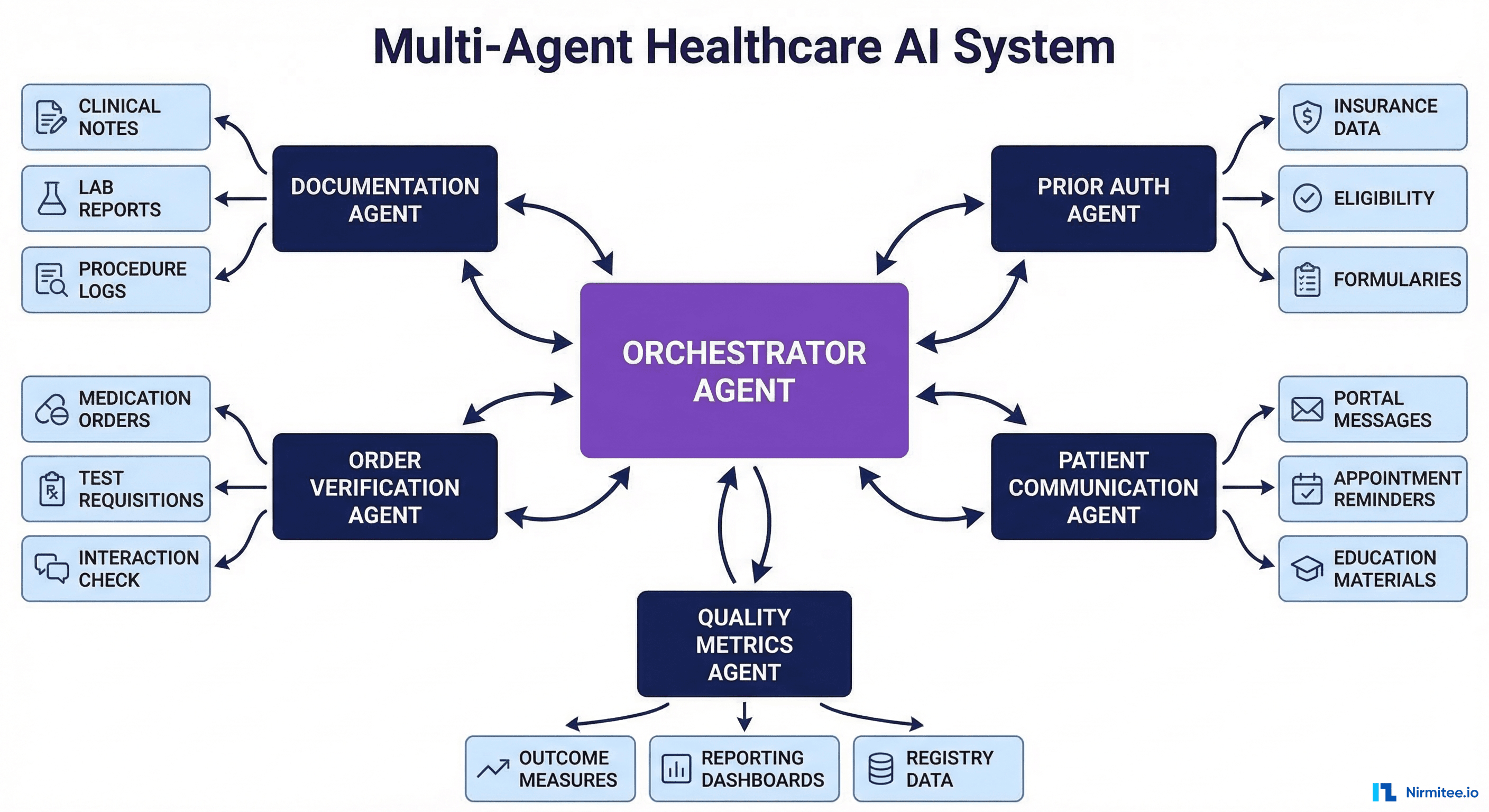
How Multi-Agent Systems Work in Clinical Settings
Consider a patient admission workflow:
- Triage Agent analyzes the chief complaint, vitals, and brief history to assign an initial acuity level.
- Documentation Agent captures the intake conversation and generates an admission note.
- Order Verification Agent reviews the admitting physician's orders against clinical guidelines, drug interactions, and clinical decision support rules.
- Prior Auth Agent identifies which ordered services require prior authorization and begins the submission process.
- Communication Agent generates a patient-friendly summary of the care plan and sends it to the patient's portal or mobile app.
Each agent operates independently but shares context through a common protocol. The orchestrator agent manages the workflow, handles failures, and ensures all agents complete their tasks in the right sequence.
The Architecture Pattern
{
"orchestrator": {
"model": "claude-3-5-sonnet-20241022",
"tools": ["route_to_agent", "check_agent_status", "aggregate_results"],
"context": {
"patient_id": "Patient/12345",
"encounter_id": "Encounter/67890",
"workflow": "admission"
}
},
"agents": [
{
"name": "documentation_agent",
"model": "claude-3-5-haiku-20241022",
"tools": ["transcribe_audio", "extract_clinical_entities", "write_fhir_document"],
"fhir_scopes": ["DocumentReference.write", "Patient.read", "Encounter.read"]
},
{
"name": "order_verification_agent",
"model": "claude-3-5-sonnet-20241022",
"tools": ["read_orders", "check_drug_interactions", "query_guidelines", "flag_alert"],
"fhir_scopes": ["ServiceRequest.read", "MedicationRequest.read", "AllergyIntolerance.read"]
}
]
}Trend 3: FHIR-Native AI Agents
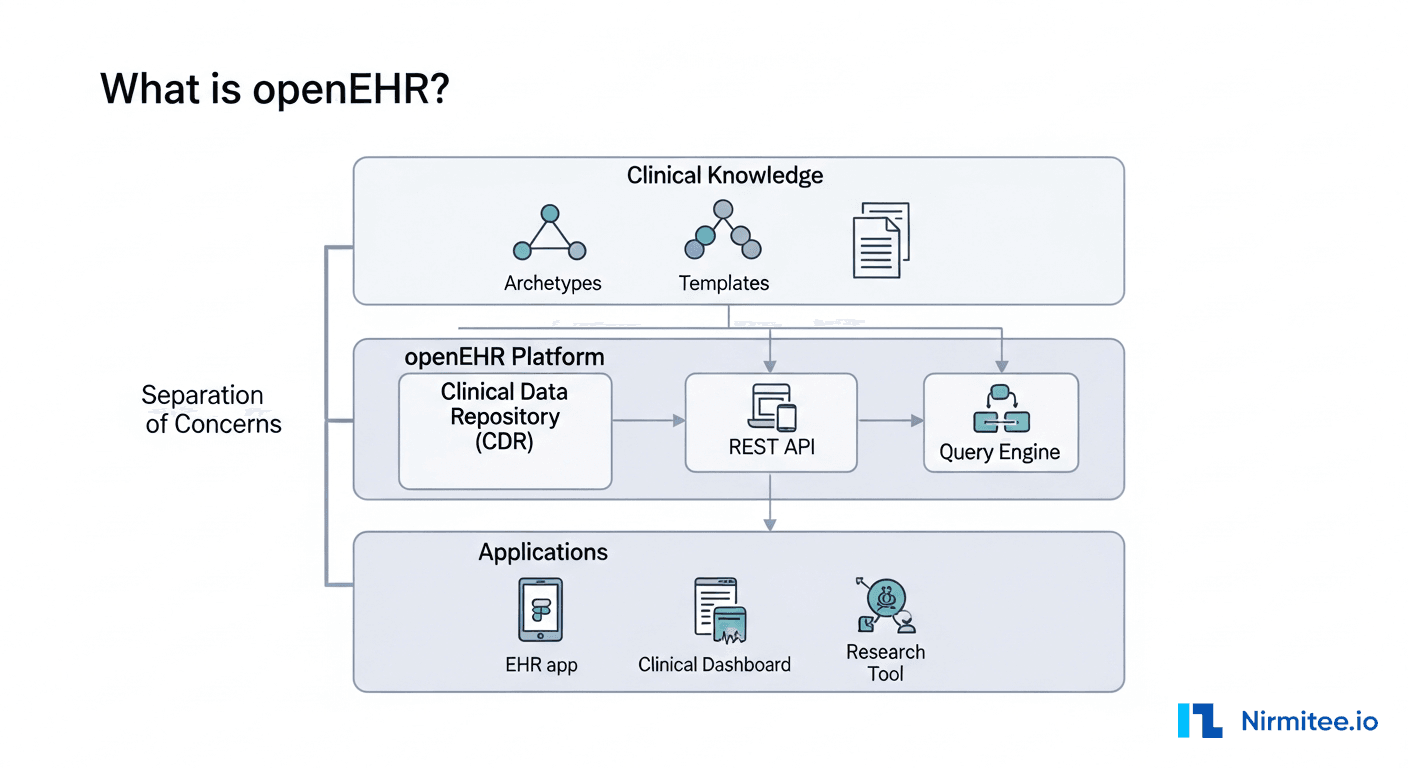
The term "FHIR-native" means an AI agent that thinks in FHIR resources, not in database rows or API abstractions. It understands what a MedicationRequest is, knows that Observation resources contain lab results, and can construct FHIR search queries to answer clinical questions.
This matters because the healthcare data model is inherently complex. An AI agent that operates on raw database tables needs custom mappings for every EHR. A FHIR-native agent works across any FHIR R4-compliant system — Epic, Oracle Health, or any open-source FHIR server.
At Nirmitee, we build FHIR-native agents using our own EHR integration infrastructure. The key technical components:
- FHIR tool definitions — Each FHIR resource type and search operation is exposed as a callable tool with typed parameters and structured responses.
- Clinical context injection — The agent receives the patient context (demographics, active conditions, current medications) at the start of every interaction, grounding its reasoning in the specific clinical scenario.
- Terminology service integration — The agent can look up ICD-10 codes, SNOMED CT concepts, and RxNorm drug identifiers to ensure clinical precision.
- Write guardrails — Any FHIR write operation requires clinician approval. The agent proposes the resource; a human confirms it.
Trend 4: Regulatory Clarity for Healthcare AI
2025 was the year regulators caught up with healthcare AI deployment. In 2026, we are seeing the first enforcement actions and the establishment of clear compliance pathways.
Key Regulatory Developments
| Regulator | Action | Impact |
|---|---|---|
| FDA | Expanded Software as a Medical Device (SaMD) framework | Clear pathway for AI clinical decision support tools |
| ONC | HTI-2 rule with AI transparency requirements | Certified health IT must disclose AI/ML usage in clinical decisions |
| HL7 International | AI Office established | Developing standards for AI model metadata, provenance, and interoperability |
| EU AI Act | High-risk classification for healthcare AI | Conformity assessment required before deployment in EU markets |
| CMS | Updated Conditions of Participation for AI-assisted care | Hospitals must have AI governance policies for CMS reimbursement |
The HL7 AI Office is particularly significant. They are developing FHIR-based standards for representing AI model metadata — including training data provenance, performance metrics, and bias assessments — as structured FHIR resources. This will standardize how AI outputs are documented in clinical records.
Trend 5: The AI Adoption Roadmap for Health Systems
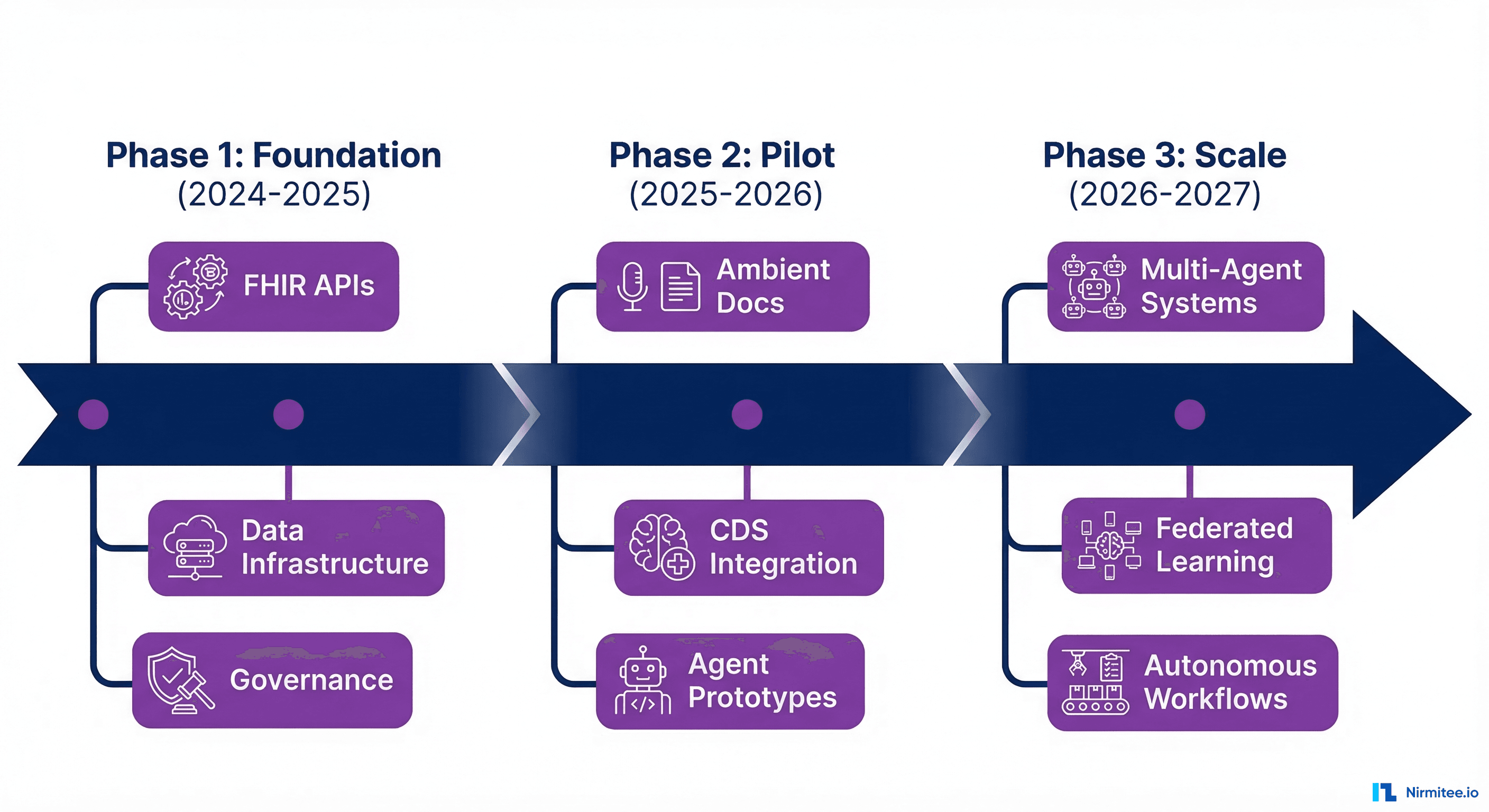
Based on our work with health systems and healthtech companies, here is the practical adoption sequence that succeeds:
Phase 1: Foundation (2024-2025)
- Build FHIR APIs — Ensure your EHR data is accessible through FHIR R4 endpoints. This is the prerequisite for everything else. Use SMART on FHIR for authentication.
- Establish data infrastructure — Implement a medallion architecture or similar data platform that provides clean, versioned datasets for AI consumption.
- Create governance frameworks — AI ethics committee, model approval process, bias assessment methodology, and incident response procedures.
Phase 2: Pilot (2025-2026)
- Deploy ambient documentation — Start with high-volume clinics where documentation burden is greatest.
- Integrate CDS models — Deploy 2-3 validated clinical prediction models (sepsis, readmission, deterioration) through CDS Hooks.
- Build agent prototypes — Develop a read-only clinical question-answering agent that retrieves data from the FHIR server to answer clinician queries.
Phase 3: Scale (2026-2027)
- Multi-agent orchestration — Connect individual agents into coordinated workflows for complex clinical processes.
- Federated learning — Participate in multi-site model training networks to improve prediction model performance.
- Autonomous workflows — Expand agent capabilities to include supervised write operations: drafting notes, suggesting orders, managing prior authorizations.
The Economic Reality of Healthcare AI in 2026
Beyond the technology trends, the economics of healthcare AI have matured significantly. Health systems and healthtech companies now have enough production data to calculate real ROI rather than relying on projections.
ROI by AI Use Case
| Use Case | Investment (Year 1) | Annual Savings | Payback Period | Source |
|---|---|---|---|---|
| Ambient documentation | $500K-$2M (vendor licensing + integration) | $1.5M-$4M (clinician time recovery) | 4-8 months | Abridge, Nuance case studies |
| Prior authorization automation | $300K-$1M | $2M-$5M (staff reduction + faster processing) | 3-6 months | Cohere Health, Waystar data |
| Clinical prediction (sepsis) | $200K-$500K | $800K-$2M (reduced mortality, shorter LOS) | 6-12 months | Epic, Johns Hopkins studies |
| Revenue cycle AI (coding) | $400K-$1.5M | $1M-$3M (fewer denials, faster reimbursement) | 5-10 months | Waystar, Optum reports |
| Patient no-show prediction | $100K-$300K | $500K-$1.5M (reduced wasted capacity) | 4-8 months | Health Catalyst data |
The pattern is consistent: healthcare AI use cases with clear workflow automation deliver positive ROI within the first year. Use cases that rely on changing clinician behavior (like predictive alerting) take longer because adoption rates are lower initially.
Vendor Landscape Shift
The healthcare AI vendor landscape consolidated significantly in 2025. Key moves:
- Microsoft acquired Nuance (completed) and embedded DAX Copilot into the Microsoft Cloud for Healthcare stack. This gives Microsoft a clinician-facing AI product that competes directly with Epic's in-house ambient documentation.
- Epic launched in-house AI across documentation, inbox management, and clinical decision support. Their strategy: build native AI features rather than relying on third-party integrations. This puts pressure on every vendor in Epic's ecosystem.
- Oracle Health (Cerner) accelerated AI integration through Oracle Cloud Infrastructure, offering AI services (OCI AI) pre-connected to Oracle Health clinical data.
- Startups consolidated — Several ambient documentation startups were acquired, and the venture capital market shifted from funding new entrants to scaling proven platforms.
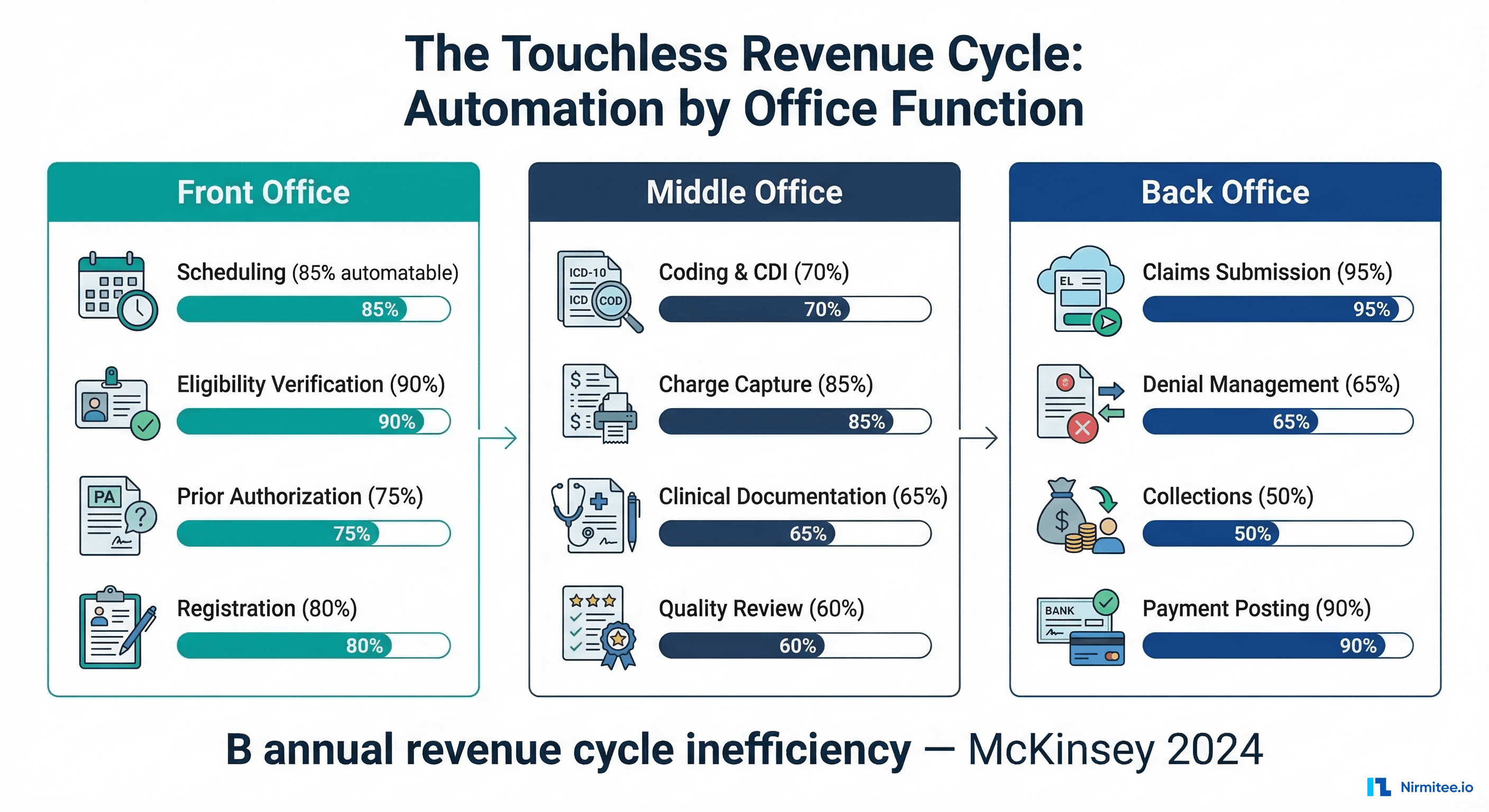
The Technical Debt Problem: Why 60% of AI Pilots Fail
According to Gartner research, approximately 60% of healthcare AI pilots do not reach production. The primary reason is not model performance — it is integration technical debt.
Common Failure Modes
- FHIR API gaps — The health system's EHR exposes FHIR endpoints, but critical data elements (like nursing assessments or medication administration records) are not available through the API. The AI model trained on complete data cannot perform when deployed against incomplete API access.
- Data quality variance — Training data was clean and standardized. Production EHR data contains free-text entries, inconsistent coding, missing values, and data entry errors. Models that achieved 95% accuracy on curated datasets drop to 70% on real-world data.
- Workflow integration friction — The AI generates valuable output, but inserting it into the clinician's existing workflow requires too many clicks or screen switches. Clinicians abandon the tool within weeks. Successful AI tools integrate into existing EHR workflows with zero or one extra clicks.
- Latency requirements — Clinical workflows have strict latency requirements. A sepsis alert that arrives 30 seconds late is useless if the clinician has already moved to the next patient. AI systems must deliver results within the clinical decision window, typically under 2 seconds for real-time use cases.
- Governance paralysis — Without a pre-established AI governance framework, every new model requires months of committee review. Health systems that establish clear evaluation criteria and approval processes before deploying their first AI model move significantly faster.
Preparing Your Stack for 2026 Healthcare AI
Whether you are a healthtech startup or a health system CTO, here is the infrastructure checklist for 2026 AI readiness:
| Infrastructure Layer | Requirement | Status Check |
|---|---|---|
| FHIR API coverage | All clinical resources accessible via FHIR R4 | Can you query Patient, Observation, Condition, MedicationRequest, DocumentReference via API? |
| SMART on FHIR auth | OAuth 2.0 + PKCE for all app integrations | Does your auth server support public client PKCE flows? |
| CDS Hooks endpoints | At least patient-view and order-select hooks active | Can external services receive hook events and return cards? |
| Data pipeline | Real-time clinical event stream available | Is clinical data flowing to an analytics platform? See our Mirth + Kafka guide. |
| Terminology services | ICD-10, SNOMED CT, RxNorm lookup available | Can your system resolve clinical codes to display names? |
| AI governance | Model approval process, bias assessment, monitoring | Do you have a documented process for evaluating and approving AI models? |
| Audit infrastructure | All AI decisions logged with provenance | Can you trace any AI output back to its input data and model version? |
What Healthtech Companies Should Do Now
- Audit your FHIR readiness — If your platform does not expose data through FHIR R4 APIs, start there. Every AI trend depends on structured, accessible clinical data.
- Choose one AI use case with clear ROI — Ambient documentation for clinician time savings, or prior auth automation for revenue cycle improvement. Ship one production use case before expanding.
- Build for multi-market compliance — The EU AI Act, FDA SaMD framework, and HL7 AI Office standards are converging. Build your AI governance infrastructure now to avoid costly retrofitting.
- Invest in integration engineering — The bottleneck for healthcare AI is not model quality. It is an integration with existing clinical workflows and EHR systems.
- Partner strategically — Healthcare AI requires clinical expertise, regulatory knowledge, and engineering depth. Very few teams have all three in-house.
Build the Future of Healthcare AI with Nirmitee
At Nirmitee, we help healthtech companies navigate the transition from AI experimentation to production deployment. Our team brings expertise in scalable healthcare architecture, AI agent development, and healthcare interoperability standards.
Contact us to discuss your 2026 AI roadmap and how we can accelerate your path to production.
Struggling with healthcare data exchange? Our Healthcare Interoperability Solutions practice helps organizations connect clinical systems at scale. We also offer specialized Agentic AI for Healthcare services. Talk to our team to get started.